|
3/15/2023 0 Comments Xef4 molecular geometry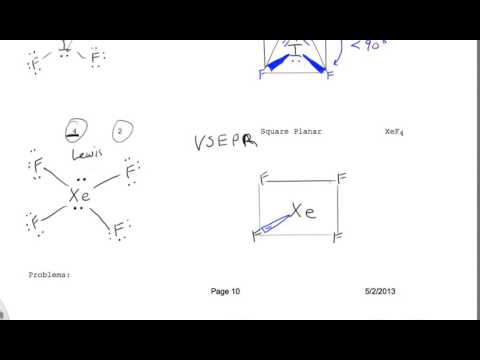 
To compute the required number of electrons that must share by each atom, F needs 8 electrons in its outermost shell and B needs most 6 valence electrons in its last shell. After computation, the result says that 24 valence electrons need to be shared to make a bond between atoms. In the compound, there is 1 atom of boron and 3 atoms of fluorine. It needs 6 valence electrons in its outermost shell. Hence, you can calculate the number of valence electrons of both the atoms using electronic configuration.Īs per the octet rule, born is an exceptional case. The atomic number of boron (B) and fluorine (F) is 5 and 9 respectively.Lone pairs (non-bonding pairs: when electrons do not participate in the formation of bonding between atoms).Required number of electrons to complete octet.To learn about any Lewis dot structure of boron trifluoride BF3, you need to compute mainly four important things. The periodic table helps you to study various elements that include atomic number, valency, etc. Sharing electrons on each atom make a covalent bond.Make sure you follow the octet rule that says each atom must complete its last shell consisting of a total of 8 electrons.At first, make a single bond with the central atom.Identify the least electronegative atom and put it in central space. 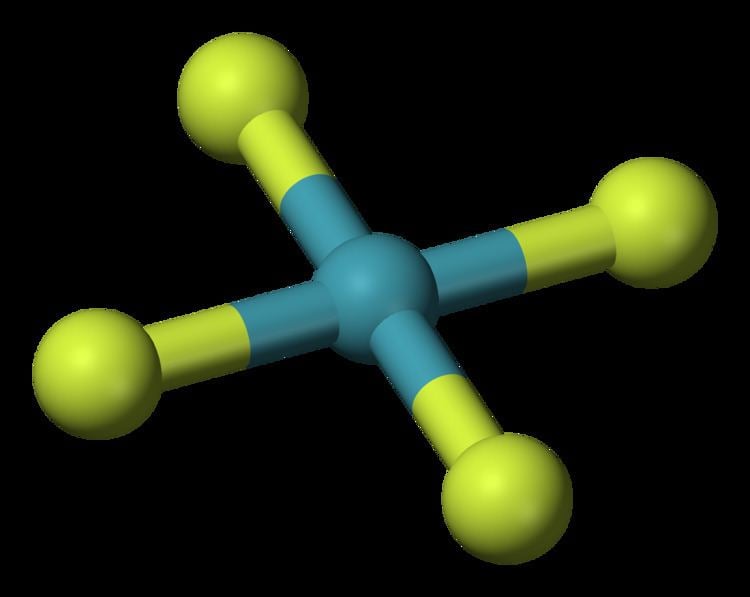
Find out the lone pairs (number of unpaired electrons) and bonding electrons (electrons that take part in the formation of a bond).Sum the product of the number of valence electrons of an atom to the number of the same atoms present in the molecule to get the total number of valence electrons of the compound.Count and calculate the number of valence electrons present in the last shell of an atom.Study the features of the periodic table that helps in finding the atomic number and informs about the electronegativity of the elements.Conclusion Points Related to Lewis Structure
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
